What is CBST Complex Sterilization?
An exclusive powder sterilization method developed through years of R&D
Introduction
- The CBST Complex Sterilization Method refers to our proprietary sterilization process developed over many years, ensuring food materials are safe from harmful bacteria.
- We supply safe, consumer-focused food materials by minimizing the destruction and alteration of nutrients through an eco-friendly sterilization method, avoiding chemical or high-temperature processes.
- It utilizes a self-developed Microwave complex sterilization system, offering a safe alternative to irradiation or chemical sterilization methods.
Why is CBST Necessary?
Stability
The Most Important Condition for Food
- Removal of harmful bacteria and viruses
- Prevention of contamination of raw materials and finished products
- Prevention of food poisoning
- Eliminating concerns about food safety
Preservation
Prevention of Food Spoilage and Deterioration
- Maintaining freshness
- Preventing changes in taste
- Preserving nutritional value
- Extending shelf life
Eco-Friendly
Sterilization is Not an Option, It is Essential
- Alternative to chemical sterilization methods
- Alternative to radiation sterilization methods
- Environmentally friendly production process
Features of The One Nature's Sterilization
Safe method using self-developed Microwave complex sterilization equipment
Non-Irradiated
No irradiation (radiation) sterilization treatment is used.
Mass Production
Mass production and bulk packaging of at least 10kg are possible.
Eco-Friendly
No harmful chemicals to the human body are used.
Non-Destructive
Minimizes the destruction of nutrients.
Self-developed Microwave Sterilization System
We supply safer and more reliable food materials through an eco-friendly sterilization process using our self-developed MICRO WAVE complex sterilization equipment, replacing irradiation or chemical sterilization methods.
How does our sterilization work?
Sterilization
Sterilization
Sterilization
Sterilization
Sterilization
Sterilization
Food materials can be contaminated throughout the entire process, from receiving to production and shipping.
The One Nature researched and tested contamination environments at each stage to develop and apply the optimal Complex Sterilization Method (CBST).
Case Study: Pathogenic Fungi Detection
※ There have been reported cases where pathogenic fungi were detected in some fermented mixed grain products.

- The importance of hygiene management for powder raw materials was confirmed based on test reports from external agencies.
- Pathogenic fungi can be effectively reduced and controlled when an appropriate sterilization process is applied.
- Systematic powder sterilization at the raw material stage is essential to ensure the safety and quality of finished products.
- Fungi and pathogenic fungi can occur due to external environmental factors during the cultivation, harvesting, drying, and storage of raw materials.
- In particular, harmful microorganisms such as pathogenic fungi and E. coli may be detected in contaminated raw materials, unhygienic production environments, or unsterilized products.
- Proactive and systematic sterilization management at the raw material stage is crucial to prevent such microbial contamination.
CBST Complex Sterilization Results
We aim for complete sterilization of various harmful bacteria such as E. coli (coliform) and pathogenic fungi, and manage the general bacteria count down to the level of 0~1,000 CFU.
← Scroll left and right →
| Test Item | Powder State (Color) | E. coli, Coliform | Pathogenic Fungi (General Bacteria) | |||
|---|---|---|---|---|---|---|
| Non-Sterilized | Sterilized | Non-Sterilized | Sterilized | Non-Sterilized | Sterilized | |
| Jujube Powder |  |
 |
 |
 |
 |
 |
| Black Sesame Powder |  |
 |
 |
 |
 |
 |
| Jerusalem Artichoke Powder |  |
 |
 |
 |
 |
 |
| Garlic Powder |  |
 |
 |
 |
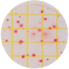 |
 |
| Anchovy Powder |  |
 |
 |
 |
 |
 |
| Beet Powder |  |
 |
 |
 |
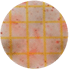 |
 |
Sterilized Packaging to Prevent External Contamination
Sterilized Packaging Standard Sizes

- Standard Packaging Unit: 10kg ~ 15kg
- Final Packaging Method:
Primary - Sterilized Packaging (PE material)
Secondary - Cardboard Box
Test & Inspection Reports

Sterilization Test Reports
Self-Quality Inspection Report
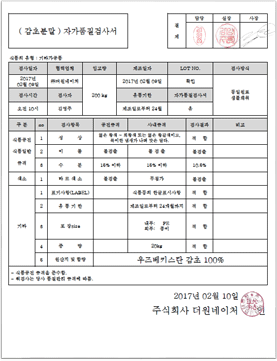
External Agency Report
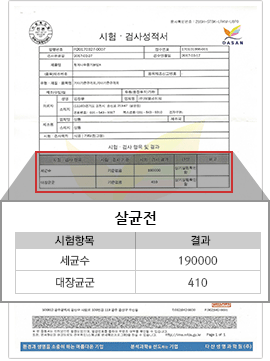
[Positive for Coliforms Before Sterilization]
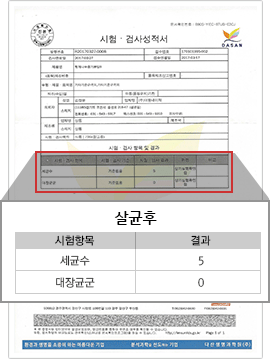
[Negative for Coliforms After Sterilization]
What is CBST+ Premium Sterilization?
Premium sterilization process optimized for preserving raw material characteristics
Introduction to CBST+ Premium Sterilization
CBST+ Sterilization is an advanced premium powder sterilization process designed to minimize changes in the material's original color and active ingredients, focusing on preserving its natural properties.
CBST Complex Sterilization vs CBST+ Premium Sterilization
You can select and apply either CBST or CBST+ depending on the raw material characteristics and product application.
| Category | CBST Complex Sterilization Method | CBST+ Premium Sterilization Method |
|---|---|---|
| Process Level |
Complex Sterilization Method
|
Premium Sterilization Method
|
| Features |
Stable sterilization and quality management
|
Enhanced minimization of color & ingredient alteration
|
| Advantages |
Thorough microbial control
|
Improved preservation of original properties
|
| Suitable Products |
Powder for pills, tablets, capsules, and sticks
|
Materials requiring color preservation
|
Case Study: Pathogenic Fungi Detection
※ There have been reported cases where pathogenic fungi were detected in some fermented mixed grain products.

- The importance of hygiene management for powder raw materials was confirmed based on test reports from external agencies.
- Pathogenic fungi can be effectively reduced and controlled when an appropriate sterilization process is applied.
- Systematic powder sterilization at the raw material stage is essential to ensure the safety and quality of finished products.
- Fungi and pathogenic fungi can occur due to external environmental factors during the cultivation, harvesting, drying, and storage of raw materials.
- In particular, harmful microorganisms such as pathogenic fungi and E. coli may be detected in contaminated raw materials, unhygienic production environments, or unsterilized products.
- Proactive and systematic sterilization management at the raw material stage is crucial to prevent such microbial contamination.
Green Tea CBST+ Sterilization Results
We aim to control various harmful microorganisms such as E. coli (coliform) and pathogenic fungi, while minimizing the color alteration of the raw materials.
← Scroll left and right →
| Test Item | Color Change | Coliform | Pathogenic Fungi | |||
|---|---|---|---|---|---|---|
| Non-Sterilized | Sterilized | Non-Sterilized | Sterilized | Non-Sterilized | Sterilized | |
| Green Tea |  |
 |
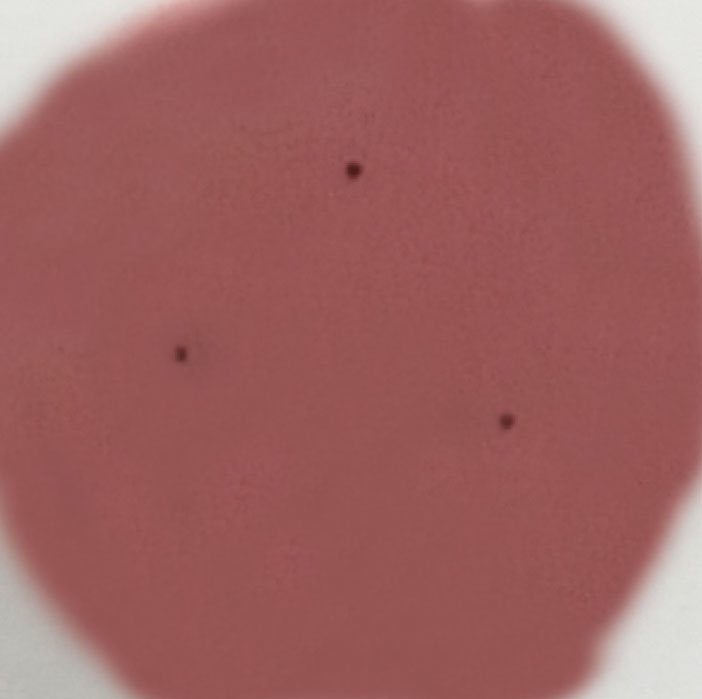 |
 |
 |
 |
※ Results may vary depending on test conditions and raw material lots.
Green Tea Component Analysis Results Before and After CBST+
Comparison of nutritional components and representative indicator component (Catechin) based on Green Tea
Preservation of Nutritional Components
Analysis of 11 ItemsMajor nutritional components remained stable with almost no change even after CBST+ sterilization.
| Category | Before Non-Sterilized |
After CBST+ |
Change |
|---|---|---|---|
| Calories (kcal/100g) | 390.24 | 390.84 | Minimal Change |
| Carbohydrates (g/100g) | 69.27 | 69.11 | Minimal Change |
| Sugars (g/100g) | 4.03 | 4.01 | Minimal Change |
| Crude Protein (g/100g) | 19.47 | 19.60 | Minimal Change |
| Crude Fat (g/100g) | 3.92 | 4 | Minimal Change |
| Saturated Fat (g/100g) | 0.31 | 0.32 | Minimal Change |
| Trans Fat (g/100g) | 0 | 0 | Maintained |
| Cholesterol (mg/100g) | 0 | 0 | Maintained |
| Sodium (mg/100g) | 4.51 | 5.13 | Minimal Change |
| Moisture (%) | 2.05 | 2.11 | Minimal Change |
| Ash (%) | 5.29 | 5.18 | Minimal Change |
※ Testing Agency : Korea Institute for Functional Foods | Results may vary depending on test conditions and lots.
Preservation of Catechin Content
Representative Indicator ComponentWe confirmed that the active ingredient, Catechin, remains stable even after the sterilization process.
| Category | Before Non-Sterilized |
After CBST+ |
Change |
|---|---|---|---|
| Catechin Content (mg/g) | 90.10 | 90.34 | Minimal Change |
※ Testing Agency : Korea Institute for Functional Foods | Results may vary depending on test conditions and lots.
🍵 What is Catechin?
Catechin is a representative functional indicator component of green tea, a polyphenol compound with excellent antioxidant and antibacterial effects. It was maintained at almost the same level (90.10 → 90.34 mg/g) even after CBST+ sterilization, confirming that sterilization does not affect the active ingredients.





